Lesson 3.5 - Thermodynamics
VIDEO LESSON (33:30 min)
Video is not accessible in some countries. If you cannot view it, you might need VPN to do so.THERMODYNAMICS
Thermodynamics is the study of heat, and its relationship to energy and work. It is a way to study energy and energy transformations in an analytical way.It is summarized by 4 general laws.

ZEROTH LAW
Two systems that are in thermal equilibrium with a third are in equilibrium with each other. We use temperature as our quantitative measure of equilibrium.
FIRST LAW
Energy can not be created or destroyed. It can only change forms.In other words, any change in a systems internal energy is equal to the heat added or work done by the system.
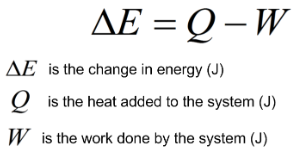
SECOND LAW
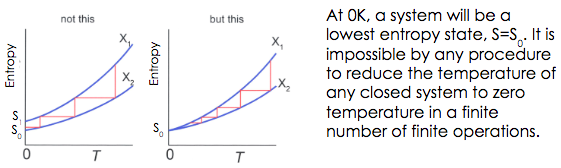
The entropy (disorder) in a closed system is always increasing.In other words, there are no completely efficient energy transformations. In every transformation there is some energy lost. It is lost as heat that increases the amount of entropy in the system.


THIRD LAW
NERNST'S THEOREM The lowest temperature is absolute zero, 0 K (kelvin) or \( -273.15^o \) C and it is impossible for a system to reach that temperature.

QUANTIFYING HEAT
How are we going to determine the amount of energy that heat is adding to a system? Heat is a measure of the motion of the atoms involved in the system. We use temperature to define this average kinetic energy.Heat is proprtional to temperature
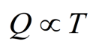
In addition, the more mass (amount of atoms) in a system the more energy is required to increase the amount of kinetic energy.
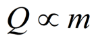
Therefore
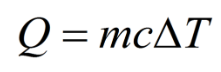
where c is the specific heat of the material. It is the amount of energy required to increase the temperature of 1 kg of the material by \( 1^o \) C.
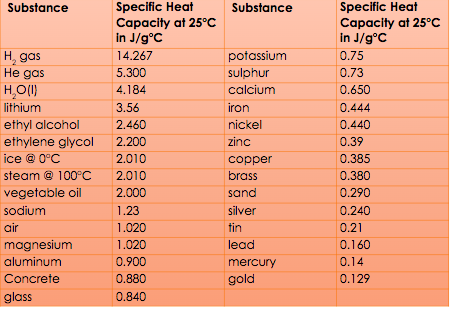
See below for the specific heat capacity for some substances.
APPLICATION
Put your hand on something metal. Now on something plastic.Question: Which is colder?
Answer: They are the same temperature. The metal has a lower specific heat. This means it takes less energy to warm it up from room temperature to the temperature of your hand (0th Law). So the rate at which it takes heat away from your is higher and it feels colder.
EXAMPLE PROBLEM #1
Determine the amount of heat required to increase 1kg of the following by \( 10^oC \):a) water
b) glass
SOLUTION:
a) \( Q=mc \Delta t \)
\(=(1kg)(4.18 KJ/Kg^oC)(10^oC) \)
\(=41.8 KJ \)
b) \( Q=mc \Delta t \)
\(=(1kg)(0.84 KJ/Kg^oC)(10^oC) \)
\(=8.4 KJ \)
EXAMPLE PROBLEM #2
A 50 g block of nickel is heated to \( 2000^o \) C. If it is dropped into 1.4 kg of water, how much does the water temperature increase?SOLUTION:
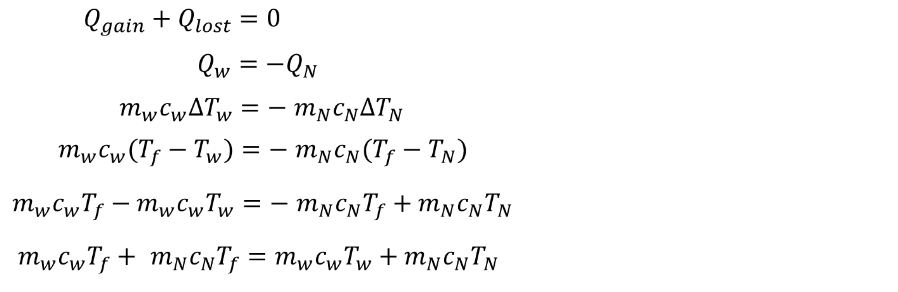

END of LESSON
Sửa lần cuối: Thứ năm, 19 Tháng 3 2026, 12:47 PM